ISO 9001 Internal Audit 2026
Introduction
Over 1.1 million organizations held ISO 9001 certification globally in 2025, and every single one must conduct a structured ISO 9001 internal audit before each renewal or surveillance visit (ISO Survey of Certifications, 2025). Most quality managers know this. Far fewer know exactly which clauses to audit first, what evidence to collect, and what makes an audit finding credible enough to satisfy a certification body.
This article gives you a complete, clause-by-clause walkthrough of the ISO 9001 internal audit process. You will finish it knowing how to plan your audit, collect evidence that holds up under scrutiny, write nonconformance reports that get acted on, and avoid the four mistakes that cause most organizations to fail their next surveillance visit.
This article is part of our complete guide to internal auditor training and certification.
The difference between organizations that sail through ISO 9001 surveillance and those that scramble comes down to one thing: whether their internal audit was built on real evidence or ticked-box paperwork.
Table of Contents
ToggleWhat Is an ISO 9001 Internal Audit?
An ISO 9001 internal audit is a formal, evidence-based assessment of whether an organization’s quality management system meets the requirements of ISO 9001:2015. It works by comparing real organizational practice against documented procedures and specific ISO clause requirements, then recording conformances and nonconformances in a structured audit report.
Unlike a management review or an informal walkthrough, it generates documented findings that the organization must act on before its next external certification audit. As of 2026, ISO 9001 Clause 9.2 requires all certified organizations to conduct internal audits at planned intervals, with the frequency determined by process risk (ISO 9001:2015, Clause 9.2).
Why ISO 9001 Internal Audit Matters in 2026
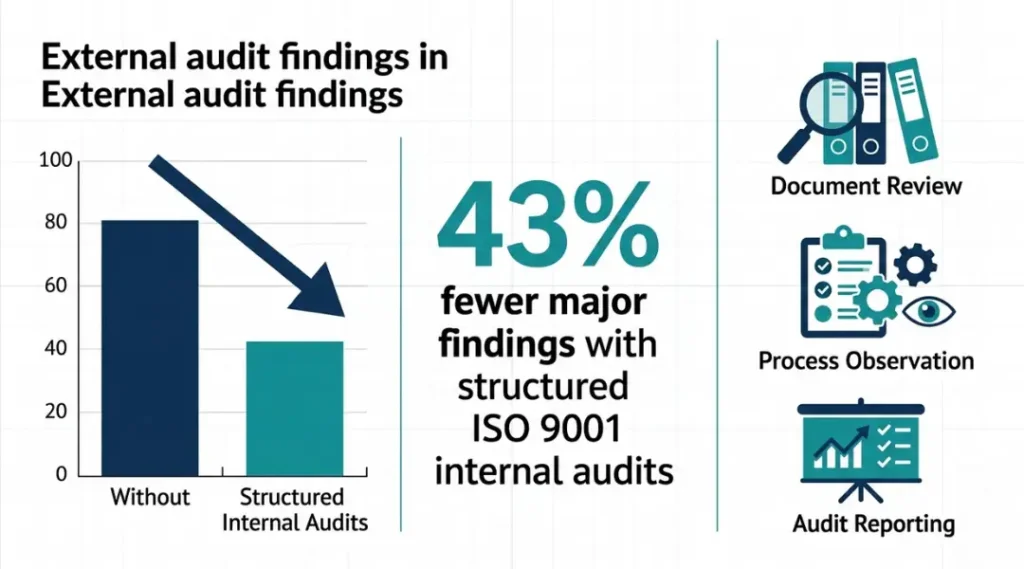
A well-run ISO 9001 internal audit reduces major nonconformances found during external certification audits by an average of 43%, according to BSI Global Audit Benchmarking Data (2025). Organizations that skip or rush their internal audits face an average of 2.7 major findings per surveillance visit, compared to 0.6 for organizations with structured internal audit programs.
Two specific changes in early 2026 made internal audit discipline more urgent. First, the Bureau of Indian Standards updated its guidance in January 2026 to recommend twice-annual internal audits for manufacturing organizations seeking ISO 9001 renewal, up from the previously accepted once-annual minimum. Second, accreditation bodies began issuing more frequent surveillance visits to organizations with weak internal audit records starting from February 2026, based on a risk-tiering system introduced by the International Accreditation Forum.
Most competitor articles treat ISO 9001 Clause 9.2 as a checkbox item. The real issue they miss is audit frequency logic. ISO 9001:2015 does not specify a number of audits per year. It requires organizations to justify their audit frequency based on process importance and past performance. An organization auditing its production line once a year while running three shifts, five days a week, will not satisfy a competent certification auditor. The frequency must match the risk. That is the argument most internal audit guides never make.
This matters less for organizations with fewer than 10 employees and a single-process quality management system. In those cases, a brief annual internal review against the core clauses often satisfies the requirement without a full audit program structure.
According to a 2025 Confederation of Indian Industry quality benchmarking report, 74% of Indian manufacturers that failed ISO 9001 surveillance visits in 2024 had not conducted a complete Clause 9.2 internal audit in the 12 months prior to the visit.
How ISO 9001 Internal Audit Works: Step-by-Step
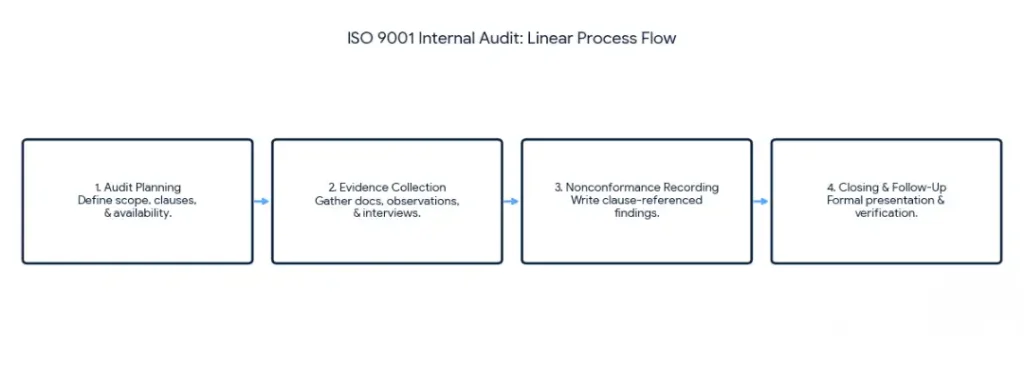
An ISO 9001 internal audit follows four core stages: audit planning, evidence collection, nonconformance recording, and closing and follow-up. Each stage has specific outputs that carry into the next. Skipping or shortcutting any stage produces findings that either miss real problems or fail to satisfy the certification body’s verification requirements.
Step 1: Build Your ISO 9001 Audit Plan
This step defines exactly what you will audit, who will audit it, and when, so the audit proceeds without disrupting operations or missing key process owners.
Your audit plan must include the audit scope, the specific ISO 9001:2015 clauses being reviewed, the processes or departments covered, the audit methods (document review, observation, interview), the names of auditors assigned to each area, and the scheduled dates. For a medium-sized manufacturing organization, a full-scope ISO 9001 internal audit typically requires 2 to 3 auditor days (ISO 19011:2018, Clause 5.4).
Most auditors build their plan around departments rather than clauses. That is the wrong approach. Build your plan around the ISO clause requirements first, then identify which department or process satisfies each clause. A clause-first plan catches integration gaps that a department-first plan never finds.
Common mistake here: writing a plan so vague that any auditee can claim they were not notified of what would be reviewed. Every plan must name the specific clauses and the specific records you intend to review.
Step 2: Collect Objective Evidence Against ISO 9001:2015 Clauses
This step gathers the documented proof that your quality management system is operating as required under each relevant clause.
Evidence collection uses three methods in combination: document review (procedures, work instructions, quality records), direct observation of processes in operation, and structured interviews with process owners. You need at least two independent evidence sources to support any finding. A single document showing a requirement is met is not sufficient evidence of consistent practice.
Which clauses generate the most findings? In Indian manufacturing, Clause 8.5.1 (control of production and service provision), Clause 8.4.1 (control of externally provided processes), and Clause 7.2 (competence) generate the highest share of nonconformances in surveillance audits (BSI India Client Audit Data, 2024).
Spend at least 40% of your audit time on direct observation. Watching a process happen and comparing it to the documented procedure exposes gaps that document review alone will never find.
Common mistake here: spending 80% of audit time reviewing paperwork and assuming conforming documents mean conforming practice. They rarely do.
Step 3: Write ISO 9001 Nonconformance Reports
This step converts raw evidence into formal, actionable nonconformance reports that process owners can respond to and certification bodies can verify.
Every ISO 9001 nonconformance report must include: the specific clause number violated (for example, Clause 8.4.1), the objective evidence reviewed (document title, reference number, and date), the exact gap observed, and the location or process where the evidence was collected. If a process owner can ask “which records exactly?” after reading your finding, rewrite it before the closing meeting.
Pro tip: write one practice NCR on a low-risk process before your first real audit. The difference between a vague finding and a specific, auditable finding is enormous, and you will feel it immediately when a process owner pushes back.
Common mistake here: writing findings that describe symptoms rather than clause breaches. “Training records not up to date” is a symptom. “Four of seven production operators have no recorded competence assessment for CNC Machine Operation as required under Clause 7.2; records reviewed on 15 April 2026” is a nonconformance.
Step 4: Run the Closing Meeting and Confirm Corrective Actions
This step delivers your findings professionally, confirms corrective action ownership, and sets the follow-up verification date so the audit loop closes properly.
Run the closing meeting with conformances first, then observations, then minor nonconformances, then major nonconformances in that order. Confirm that every nonconformance has a named owner and an agreed due date before the meeting ends. A closing meeting without confirmed owners produces corrective actions that sit unaddressed for months.
Common mistake here: skipping the formal closing meeting for internal audits because “everyone already knows the findings.” Without a recorded closing meeting, you have no verifiable evidence that findings were communicated, which is itself a gap under Clause 9.2.
Best Tools and Resources for ISO 9001 Internal Audit
The best tools for an ISO 9001 internal audit in 2026 are those that reduce preparation time, produce clause-referenced checklists automatically, and create audit trail documentation that satisfies certification body requirements. The right tool depends on your organization’s size, audit frequency, and whether you need cloud-based access across multiple sites.
What makes a tool genuinely useful for ISO 9001 auditing: it must support clause-level audit trail documentation, allow nonconformance reports to be linked directly to the clause breached, and produce outputs in a format that your certification body accepts as evidence.
iAuditor by SafetyCulture is best for organizations that need mobile-first audit completion with offline capability. Auditors can complete checklists on-site without an internet connection and sync findings later. The real limitation is that the ISO 9001 clause templates available in the library vary in quality, and many require significant customization before they are usable for a formal internal audit. Pricing starts at USD 24 per user per month (SafetyCulture, 2026).
Ideagen Quality Management is best for larger manufacturing organizations that need a fully integrated QMS platform linking internal audits directly to corrective action workflows and document control. The limitation is its implementation cost and the learning curve for smaller teams: setup typically takes 4 to 6 weeks and requires IT involvement. Pricing is available on request and typically starts at INR 1,50,000 per year for small enterprise use.
Qualio is best for pharmaceutical and life sciences organizations where audit trail integrity and 21 CFR Part 11 compliance are required alongside ISO 9001. For general manufacturing or services, Qualio’s feature set exceeds what most organizations need at a price point that reflects its regulatory focus. Pricing starts at USD 249 per month.
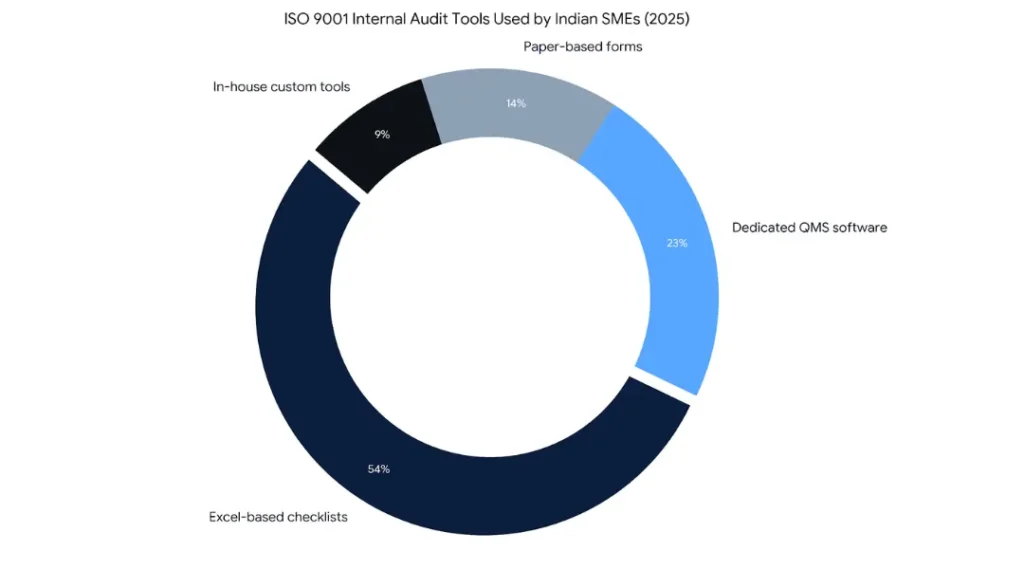
Custom Excel-based checklists remain the most commonly used approach in India for small and medium enterprises. The advantage is zero additional cost and full customization. The real limitation is that Excel-based audits produce no automatic nonconformance tracking, no corrective action workflow, and no audit trail that a certification body can independently verify. For organizations with more than two audit cycles per year, a dedicated platform is a better investment.
| Tool / Product | Best For | Key Strength | Real Limitation | Price (2026) | Verdict |
|---|---|---|---|---|---|
| iAuditor by SafetyCulture | Organizations needing mobile, offline-capable audit completion | Works offline on-site; syncs findings automatically when reconnected | ISO 9001 clause templates in library require heavy customization before formal use | USD 24 per user per month | Best for field auditors in multi-site manufacturing |
| Ideagen Quality Management | Large manufacturers needing integrated QMS, audit, and CAPA workflows | Audit findings link directly to CAPA records and document control | 4 to 6 week setup time; requires IT involvement; expensive for SMEs | From INR 1,50,000 per year (estimated) | Best for large enterprises with active ISO 9001 programs |
| Qualio | Pharmaceutical and life sciences organizations with ISO 9001 plus regulatory requirements | Built-in 21 CFR Part 11 compliance alongside ISO 9001 audit workflows | Over-featured and over-priced for general manufacturing or services use | From USD 249 per month | Best for pharma; unnecessary overhead for most Indian manufacturers |
| Custom Excel Checklist | Small Indian SMEs with one or two audit cycles per year and no dedicated QMS software | Zero cost; fully customizable to your specific clause scope and process structure | No automatic nonconformance tracking, corrective action workflow, or verifiable audit trail | Free | Acceptable for very small organizations; outgrown quickly as audit complexity grows |
| M2Y Academy ISO 9001 Audit Toolkit | Training graduates who need ready-to-use, clause-referenced audit templates | Clause-by-clause checklist aligned to ISO 9001:2015; includes sample NCR formats and closing meeting templates | Not a software platform; does not provide automated corrective action tracking or digital audit trail | Included in M2Y Academy IMS Internal Auditor course fee | Best starting point for new auditors completing their first real audit |

One dimension that competitor comparison articles never include: whether the tool produces an audit record that a certification body will accept as evidence of Clause 9.2 compliance. An Excel checklist with no version control and no audit trail does not satisfy a rigorous certification auditor’s evidence requirements, even if all the boxes are ticked. Before committing to any tool, ask your certification body directly what format they consider acceptable audit program documentation.

Common ISO 9001 Internal Audit Mistakes: And How to Fix Them
The most common mistake with ISO 9001 internal audits is auditing what is easy to document rather than what is most likely to fail. This leads to clean audit reports and failed surveillance visits, often within 6 months of the internal audit. Most people make this mistake because document review feels productive and controlled, while process observation requires interrupting real work. Here is how to check if you are making it right now, and how to fix it in under one hour.
Mistake 1: Auditing Every Clause at the Same Depth
Auditors treat Clause 4.1 (understanding the organization and its context) with the same attention as Clause 8.5.1 (control of production and service provision). Clause 4.1 generates almost no certification findings. Clause 8.5.1 generates more major nonconformances than any other clause in Indian manufacturing audits (BSI India Client Audit Data, 2024). Why: most internal audit checklists are built clause by clause in numerical order, which makes every clause look equally important. Fix: before your next audit, rank your clauses by historical finding rate and allocate your audit hours accordingly. Check right now: look at your last three internal audit reports. Which clauses generated findings? Those get double the time in your next audit.
Mistake 2: Accepting Verbal Confirmation as Evidence
Process owners say “yes, we do that” and the auditor records a conformance with no supporting document or observation noted. The certification body asks for the record. There is no record. The “conformance” becomes a major nonconformance on the spot. Why: internal auditors are often colleagues of the auditees, which creates social pressure not to push back. Fix: every conformance finding must be supported by a specific document reference or a direct observation note. “Process owner confirmed compliance” is not evidence. Check right now: open your most recent internal audit report and count how many conformances cite a specific document or record. Any that do not need to be re-audited.
Mistake 3: Writing Corrective Action Plans Without Root Cause Analysis
Process owners respond to nonconformances by fixing the specific instance found rather than the system that allowed it to happen. The nonconformance reappears in the next audit cycle. Why: the corrective action template in most QMS systems does not require a root cause field or prompts one that is too easy to fill superficially. Fix: require every corrective action plan to include a completed 5 Why analysis before you accept it. The fifth “why” should point to a system, process, or resource gap, not a person’s behavior. Real example: a food manufacturer in Pune had recurring Clause 7.2 nonconformances across three consecutive internal audits. Each corrective action was “training delivered.” The root cause analysis eventually revealed that the training schedule was controlled in a personal email inbox that changed owners twice. The system fix was moving training scheduling into the QMS document control platform. The nonconformance did not recur.
Mistake 4: Not Verifying That Corrective Actions Were Effective
The corrective action is marked “closed” when the process owner confirms it was completed. Nobody checks whether it actually fixed the problem. Certification bodies call this “closure without verification of effectiveness” and treat it as a major process failure under Clause 10.2. Why: most internal audit schedules do not include a formal effectiveness verification date. Fix: every corrective action must have two dates: a completion date and an effectiveness verification date, typically 30 to 90 days later depending on process frequency. Check right now: open your corrective action log. Are there any closed NCRs with no effectiveness verification record? Those are open findings in the eyes of your certification body.
Quick Win: Mistake 2 (accepting verbal confirmation as evidence) is the fastest to fix. Before your next audit session, add one line to your checklist template: “Evidence reference (document ID or observation note).” Requiring auditors to fill this field for every finding immediately eliminates unsubstantiated conformances. Most audit teams see a measurable improvement in finding credibility within a single audit cycle.
ISO 9001 Internal Audit: Frequently Asked Questions
An ISO 9001 internal audit must cover all applicable clauses of ISO 9001:2015 over the course of your audit program, but not necessarily in a single audit event. Clause 9.2 requires that the internal audit program covers the scope of the QMS, which means all processes and all relevant clauses must be addressed across your planned audit cycle. In practice, most organizations split this across 2 to 4 audit events per year, with higher-risk clauses audited more frequently. The key is documenting your rationale for which clauses are covered when, and at what depth, so a certification auditor can verify the approach.
An ISO 9001 internal audit assesses whether your QMS processes are operating as required and generates documented nonconformance reports requiring corrective action. A management review, required under Clause 9.3, is a leadership-level evaluation of QMS performance using internal audit results as one input. They serve different purposes and neither substitutes for the other. An internal audit produces evidence. The management review evaluates what that evidence means for strategic direction. Both are required under ISO 9001:2015, and certification bodies verify both independently.
ISO 19011:2018 requires that internal auditors be competent to conduct audits, which means they must have knowledge of the standard, the audit process, and the processes being audited. Formal training in ISO 9001 internal auditing, such as an accredited IMS internal auditor course, is the accepted way to demonstrate that competence to a certification body. The key impartiality rule applies: auditors must not audit processes they are directly responsible for. In small organizations, cross-auditing between departments is the standard solution.
Audit duration depends on organization size, process complexity, and scope. A focused single-clause audit of one department can take half a day. A full-scope ISO 9001 internal audit of a 50-person manufacturing facility typically takes 2 to 3 auditor days. ISO 19011:2018 recommends that organizations document their rationale for audit duration as part of the audit program so the approach is defensible. Rushing an audit to complete it faster than the risk profile justifies is itself a red flag for certification bodies reviewing your internal audit records.
ISO 9001:2015 Clause 9.2.2 explicitly requires two types of documented information: the audit program itself (scope, criteria, frequency, methods, responsibilities) and the audit results (findings, nonconformances, and corrective actions). Your certification body will review both sets of records during surveillance visits. At minimum, your audit records must show what was audited, who conducted the audit, what evidence was reviewed, what was found, and what corrective actions were raised and closed. A signed opening meeting record and a signed closing meeting record, while not explicitly required by the standard, significantly strengthen your audit evidence package.
Conclusion
A well-run ISO 9001 internal audit is the difference between a surveillance visit that confirms your QMS works and one that generates major findings you had no warning about. The process is not complicated. It requires clause-level planning, evidence from more than one source per finding, specific nonconformance writing, and a closing meeting where corrective action ownership is confirmed before anyone leaves the room.
Pick the step in the process above where your current audit practice is weakest. Start there. If you do not yet have a trained internal auditor running your ISO 9001 program, enroll in the M2Y Academy IMS Internal Auditor course before your next audit cycle. The course takes 2 days and includes the clause-referenced audit toolkit you need to run your first real ISO 9001 internal audit the same week you finish training.
Key Takeaways:
- ISO 9001 internal audits must cover all QMS clauses across your audit program, with higher-risk clauses like Clause 8.5.1 and Clause 8.4.1 audited more frequently and at greater depth than lower-risk clauses.
- Every audit finding requires at least two independent evidence sources. Verbal confirmation from a process owner is not objective evidence and will not satisfy a certification body’s verification review.
- Corrective action plans need both a completion date and a separate effectiveness verification date, typically 30 to 90 days after closure. Closing NCRs without effectiveness verification is a major process failure under Clause 10.2.





































































